|
However, recent studies suggest that exogenous citrate appreciably contributes to intermediary metabolism only in oxygen- and glutamine-limited conditions. This last pool of citrate also depends on the activity of a plasma membrane citrate transporter which catalyzes the entry of external citrate into the cell. The CIC is therefore able to modulate the amount of citrate both in the mitochondrial matrix and in the cytosolic space. It is firmly inserted in the inner mitochondrial membrane where it catalyzes with high affinity the flux of citrate and other substrates such as isocitrate across the lipid bilayer in strict cooperation with many catabolic and anabolic reactions occurring inside and outside mitochondria. The mature protein is made up of three homologous sequence domains, each of them containing ~ 100 amino acids and organized in the form of 2 α-helices. The protein is synthesized in the cytosol as a precursor protein with an amino-terminal presequence of 13 amino acids whose role has been extensively investigated. The human CIC is encoded in the nucleus by the SLC25A1 gene located on chromosome 22q11.2. Then, the protein was purified from rat and bovine liver, yeast, eel and maize mitochondria and functionally reconstituted into liposomes to study its transport properties. The mitochondrial CIC (SLC25A1) was discovered in 1967 by Chappell and Haarhoff in rat liver mitochondria. By doing so, the CIC links intra-mitochondrial and extra-mitochondrial (cytosolic) reactions in which the substrates transported by this carrier protein are involved. In this complex metabolic network, the citrate carrier (CIC) mediates the efflux of citrate and other metabolites from the mitochondrial matrix to the cytosol. Therefore, the transport activities of these proteins are carefully orchestrated to channel several molecules belonging to the same metabolic pathway in a fine and coordinated manner. 
By transporting several substrates, these proteins are involved in a variety of biochemical processes, such as Krebs cycle, oxidative phosphorylation (OXPHOS), the transfer of reducing equivalents, gluconeogenesis, fatty acid metabolism, amino acid synthesis, and cofactor transport. 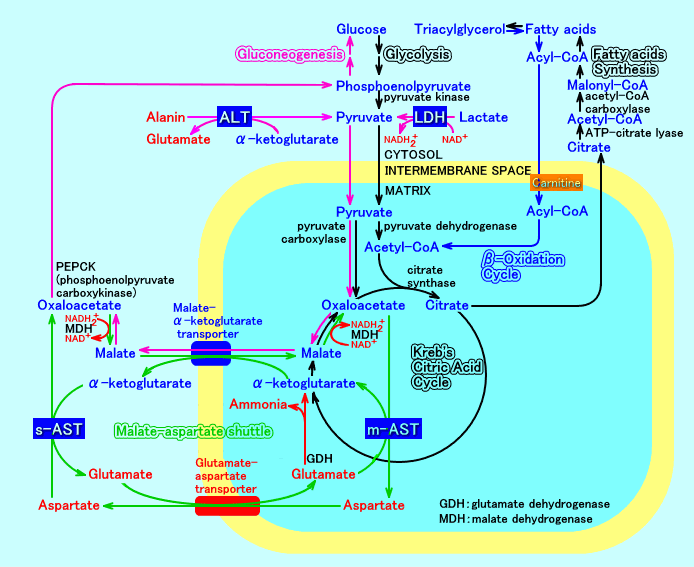
Most of these metabolite carriers, encoded by the nuclear genes of the SLC25 family, have been characterized both structurally and functionally in several species. Mitochondrial carrier proteins play an essential role in the control of the flow of metabolites between the cytosol and mitochondrial matrix, thus contributing to many cellular processes. Finally, the metabolic significance of the citrate flux, mediated by the CIC, across distinct subcellular compartments is also discussed. In this review, we critically analyze these data starting from the multiple roles played by the mitochondrial CIC in many cellular processes and then examining the regulation of its activity in different nutritional and hormonal states. A lot of data on the characteristics of this protein have therefore accumulated over time thereby leading to a complex framework of metabolic and physiological implications connected to the CIC function. Several studies have been carried out over the years on the structure, function and regulation of this metabolite carrier protein both in mammals and in many other organisms. In this way, the CIC provides the cytosol with citrate which is involved in many metabolic reactions. 
The citrate carrier (CIC) is an integral protein of the inner mitochondrial membrane which catalyzes the efflux of mitochondrial citrate (or other tricarboxylates) in exchange with a cytosolic anion represented by a tricarboxylate or a dicarboxylate or phosphoenolpyruvate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
